Radium-223 in the Treatment of Osteoblasticmetastases a Critical Clinical Review

Radium-223-Dichloride in Castration Resistant Metastatic Prostate Cancer—Preliminary Results of the Response Evaluation Using F-18-Fluoride PET/CT
1
Departments of Molecular Radiotherapy & Nuclear Medicine, Docrates Cancer Center, Saukonpaadenranta 2, Helsinki FI-00180, Finland
2
Departments of Radiotherapy and Medical Oncology, Docrates Cancer Center, Saukonpaadenranta ii, Helsinki FI-00180, Republic of finland
*
Author to whom correspondence should be addressed.
†
Present Accost: Department of Nuclear Medicine, Academy of Texas MD Anderson Cancer Center, 1400 Pressler, Houston, TX 77030, USA.
Academic Editor: Stephen Assinder
Received: 21 Baronial 2015 / Revised: 25 September 2015 / Accustomed: 29 September 2015 / Published: 13 Oct 2015
Abstract
The purpose of this study was to evaluate the outcome afterward Radium-223-dichloride (223RaClii) treatment of patients with skeletal metastases of castration resistant prostate cancer using whole-body 18F-Fluoride PET/CT. Sodium eighteenF-fluoride [eighteenF]-NaF PET/CT was performed prior the treatment of 223RaCltwo, subsequently the kickoff cycle and later the 6th cycle. The skeletal metastases were analyzed quantitatively using modified PET response evaluation PERCIST criteria. The patients were also analyzed for South-PSA. All x patients responded in [18F]-NaF scans afterwards vi cycles, but interim analysis after the 1st cycle did not give additional information almost the event. The S-PSA decrease correlated with [xviiiF]-NaF response, but 1 patient demonstrated progressive disease, i.e., >25% increase in S-PSA values during 223RaCl2. Our results (although preliminary) advise that 18F-Fluoride PET/CT is useful in the follow-upwardly of castration resistant prostate cancer with skeletal metastases.
1. Introduction
Prostate cancer is the virtually common cancer in Europe and 10%–20% of patients present with avant-garde or metastatic affliction with associated symptomatic bone metastases [1]. Bone is the near frequent metastatic site in prostate cancer, with approximately 90% of patients with metastatic castration resistant prostate cancer (mCRPC) having radiological evidence of bone metastases [ii]. Bone metastases profoundly affect an private'south quality of life, increase the risk of bone marrow failure [iii] and skeletal-related events (SREs) such as pathological fractures and spinal cord compression [4], and significantly reduce life expectancy [5,6]. The main cause of disability and expiry amongst those with mCRPC is bone metastases.
According to EANM guidelines, patients considered for therapy with beta-emitting radiopharmaceuticals such as 89Sr-Cl, 153Sm-EDTMP or 186Re-HEDP therapy had to fail in conventional analgesics and anti-tumor therapy, chemotherapy or hormone therapy. Pain had to be severe enough to limit normal activities and/or require regular analgesics, and patients had to undergo recent (within 8 weeks or less) bone scintigraphy documenting increased osteoblastic activity at painful sites. These radionuclide treatments are palliative, where sixty%–80% of patients benefit from the handling and they do not cure metastatic cancer [7,8]. All of these agents take demonstrated improved overall survival in retrospective controlled studies [9,10,11], but at that place are non even so randomized controlled studies which back up these findings, but studies which do not improve survival [12]. In 2010'southward, the bone-seeking radiopharmaceutical, radium-223 dichloride (223RaCl2), was introduced into clinical practise. 223RaCl2 is indicated for the handling of patients with mCRPC and symptomatic bone metastases, offering overall survival benefits comparable to the other new treatment options available [13,fourteen,15]. The availability of 223RaCl2 has generated considerable interest amongst the nuclear medicine community, and in many centers around the globe nuclear medicine physicians take joined the multidisciplinary treatment squad involved in the management of patients with mCRPC.
This 223RaCl2 is an alpha-emitting bone targeting agent, which has favorable pharmacokinetic and pharmacodynamic characteristics: at 10 min 12%, at 1 h half dozen% and at 24 h <one% of the injected activity is in the blood and the skeletal uptake is 44%–77% at 4 h. Fecal excretion is the major elimination route, but additionally 5% is excreted in the urinary tract [fifteen,xvi]. In regards to diagnostic imaging of these patients, the Social club for Nuclear Medicine published guidelines for [18F]-NaF PET/CT bone scans [17]. Bone uptake of xviiiF-NaF reflects bone remodeling, and it is part of the mineralization of os matrix. [18F−] ion is exchanged for [OH−] then that hydroxy apatite bone matrix is transformed into fluoro apatite, indicating that high uptake of [xviiiF]-NaF reflects bone reactions to os injury, not to prostate cancer. Positive findings with [18F]-NaF PET/CT are due to both beneficial and malignant bone disorders. In the literature, x articles described the use of [18F]-NaF PET/CT scans in the diagnosis of metastatic prostate cancer [xviii,19,20,21,22,23,24,25,26,27]. In our ain meta-assay most [eighteenF]-NaF PET/CT in 3918 patients [28], 1289 (33%) had positive scans. Use of [18F]-NaF and xviiiF-Choline PET/CT had like diagnostic accuracy at staging for patients with prostate cancer just 18F-Choline has college specificity at restaging for recurrence [28].
In this article, the consequence of 223RaClii treatment was analyzed using [18F]-NaF-PET.
2. Methods and Patients
Patients: Prostate cancers were diagnosed between 2001 and 2013. In this retrospective one plant analysis we included all those patients who had received at least vi cycles of 223RaCltwo therapy. Men'south ages ranged from 49 to 83 years; Gleason scores were 6–10; initial PSA values varied from five.5 to 15,500 μg/L, and all patients had T3-T4 disease. All the patients had skeletal metastases, and two of them additionally had visceral metastases. The radiological TNM staging before 223RaCltwo was performed using [18F]-NaF-PET and [18F]-fluorocholine-PET in all patients, almost (9 out of 10) of the patients had too pelvic MRI and diagnostic whole body CT. The 223RaCl2 injections were given every 4 weeks and follow up studies were washed with [18F]-NaF. Two of them had previous surgery, 8 had previous radiotherapy, all 10 had androgen impecuniousness therapy (ADT) besides having received chemotherapy, 2 had received Sm-153-EDTMP therapy, and nine additionally received denosumab prior 223RaCl2. The patient characteristics are summarized in Table 1.
This work is a retrospective treatment analysis of our ten kickoff Ra-223 patients followed with [eighteenF]-NaF PET/CT imagings. This retrospective analysis was performed, co-ordinate to the principles of the Declaration of Helsinki and our patient database was canonical by the Finnish authorisation for the protection of privacy and personal information. All patients gave a written informed consent for the employ their information.
Imaging PET/CT protocol: Imaging was done on Siemens Biograph PET Scanner, combined with depression-dose CT. The injected activeness dose of [18F]-NaF ranged 212–290 MBq. Whole trunk imaging was performed by starting at 60 ± 3.0 min (range 58–76 min) from the calvarium to the tips of the toes using 2.five min per bed position. All analyzed patients were treated with half-dozen cycles of radium-223-dichloride (223RaCl2) and [18F]-NaF PET/CT imaging was practical before Ra-223 therapy in all patients, in 6 patients afterwards the 1st cycle and in all patients later on the terminal sixth cycle (Tabular array ii). In i patient (no. 6) at that place was a production failure of 223RaClii and the interval betwixt the 1st and 2nd cycle was 10 weeks instead of normal 4 weeks. He was imaged after the 2nd wheel, and received a total of seven cycles. The [18F]-NaF later on the first (second) cycle was performed 3 weeks after 223RaClii infusion. The last control was performed within 4 weeks after the last 6th (7th) wheel in all patients. The tracer [18F]-NaF was provided from MAP Medical Technologies Oy (Helsinki, Republic of finland).
Blastoff-therapy: 223RaCl2 (Xofigo®) was caused from Bayer Healthcare AG (Berlin, Germany), each single-apply vial contained half-dozen mL of solution at a concentration of yard kBq/mL (27 μCi/mL) and a total radioactivity of vi MBq/vial (162 μCi/vial) at the reference date [29]. The used activity dose of 223RaCl2 is fifty kBq/kg body weight (1.35 μCi/kg) given at 4-weekly intervals for six cycles, the cumulative activities are listed in Table two. The volume administered was calculated using the patient's body weight (kg), the dosage level (l kBq/kg trunk weight), the radioactivity concentration of the product at the reference appointment, and the decay correction factor provided with each vial.
Prior to the get-go dose, patients were analyzed for a full blood count (FBC) to assess bone marrow part. Treatment was given if: haemoglobin level >10 g/dL, absolute neutrophil count (ANC) ≥1.v × 109/50, and platelet count ≥100 × 10nine/L. Due to the potential myelotoxic effects of handling, a FBC was undertaken prior to all subsequent handling cycles. The conclusion to administer the adjacent cycle depended on recovery of the ANC and platelet counts to levels of ≥one.v × 109/L and ≥100 × 109/L, respectively. The cumulative doses in 6 cycles varied from 16.45 to 31.57 MBq.
Table 1. The patient characteristics.
| Patient Age | Diagnosis Gleason Score, TNM (Yr) | Distribution (earlier Ra-223) | Dx/modality * | Cycles * Cumulative Activeness | Initial S-PSA | Other Treatments before Xofigo |
|---|---|---|---|---|---|---|
| 1 m/63 | GS 9, T4N1M1 (-13) | Prostate Os lnn | NaF, FCH, MRI, CT | 6 30.86 MBq | 410 | Degarelix, Bicalutamide, Docetaxel, Abiraterone, Enzalutamide, Denosumab |
| 2 thousand/77 | GS vii, pT3N0M0 (-01) | Bone | NaF, FCH, FACBC, MRI, CT | 6 24.80 MBq | 5.5 | RRP, EBRT (PO), Bicalutamide, Docetaxel, Abiraterone, Enzalutamide, Denosumab, Goserelin |
| iii m/68 | GS 8, T4N1M1 (-thirteen) | Bone | NaF, FCH, MRI, CT | half dozen 27.08 MBq | 15,500 | Degarelix,Bicalutamide, Denosumab, EBRT (B), Docetaxel, Abiraterone, EBRT (P), Enzalutamide, Denosumab, Goserelin |
| 4 yard/49 | GS 9, T4N1M1 (-13) | Bone | NaF, FCH, MRI, CT | 6 31.57 MBq | 700 | Cyproterone, Degarelix, Bicalutamide, Zoledronate, Docetaxel, Sm-153-EDTMP, Mitoxantrone, EBRT (P), Docetaxel, Abiraterone |
| five m/67 | GS nine, T3N1M1 (-13) | Os | NaF, FCH, MRI, CT | 6 25.87 MBq | 430 | Degarelix, Bicalutamide, Denosumab, Docetaxel, Goserelin, Enzalutamide |
| vi k/69 | GS 8, T4N1M1 (-11) | Bone | NaF, FCH, MRI, CT | 7 24.13 MBq | 16.2 | Bicalutamide, Goserelin, EBRT (P), Abiraterone, Denosumab, Docetaxel, Enzalutamide |
| 7 m/56 | GS 10, T4NxM1 (-13) | Os | NaF, FCH, MRI, CT | 6 22.78 MBq | 790 | Bicalutamide, Lupron, Denosumab, EBRT (P), Sm-153-EDTMP, Mitoxantrone, Docetaxel, Abiraterone |
| viii yard/58 | GS 9, T3N0M1 (-12) | Bone | NaF, FCH, MRI, CT | 6 23.71MBq | 6.5 | Denosumab, Degarelix, Bicalutamide, EBRT (P), Goserelin, Docetaxel |
| 9 m/lx | GS 9, T4M1N1 (-12) | Bone | BS, NaF, FCH, MRI, CT | 6 sixteen.45 MBq | 18 | Bicalutamide, Buserelin, TURP, Docetaxel, EBRT (B), Abiraterone, Denosumab, Cabazitaxel, Enzalutamide |
| ten m/83 | GS 6, T3NxM0 (-01) | Bone Liver lnn | NaF, FCH | half-dozen 24.06 MBq | eighteen | Leuprolin, EBRT (P), Docetaxel, Abiraterone, Mitoxantrone, Zoledronate, Denosumab |
Paradigm analysis: Lesions were considered aberrant when focal tracer accumulation was greater than background activity, usually if the SUV values were higher than 10.
Interpretation of bone lesions (benign or malignant) depended on anatomical localization; and the five regions: skull, vertebral column, thoracic girdle, pelvic girdle and extremities were estimated quantitatively using modified PERCIST criteria for NaF. In the archetype PERCIST assay, the skeleton is considered as organ, and a maximum of 2 lesions per organ are taken into business relationship out of the total sum of five lesions. Therefore, here two highest SUVmax values of skeletal uptakes from two regions were summed. The results were compared with those of base line, the alter of 6% were considered significant as in conventional PERCIST consisting of v lesions, where xv% change is considered pregnant which is analogous to PERCIST criteria for early response [30].
Tabular array 2. The summary of the NaF imaging results.
| Patient Age | Bone Distribution # (before Ra-223) | NaF-% Change later 1st/ 6th Cycle (2nd/seventh) | Number of Cycles Cumulative Activeness | S-PSA Response | Treatments during Xofigo |
|---|---|---|---|---|---|
| one m/63 | Bone 3/3 | +0.8/−thirteen.8 | 6 30.86 MBq | PD | Degarelix, Enzalutamide, Denosumab |
| two chiliad/77 | Bone 2/3 | +half dozen.2/−6.9 | 6 24.80 MBq | PR | Enzalutamide, Denosumab, Goserelin |
| 3 g/68 | Os 3/3 | −10.8/−9.2 | vi 27.08 MBq | CR | Enzalutamide, Denosumab, Goserelin |
| four chiliad/49 | Bone iii/3 | nd/−thirteen.0 | 6 31.57 MBq | PD/SD | Lupron,Denosumab, Abiraterone |
| 5 m/67 | Bone 1/3 | −6.5/−7.eight | half dozen 25.87 MBq | PR | Denosumab, Goserelin, Enzalutamide |
| 6 chiliad/69 | Bone iii/3 | −x.4/−11.vii | seven 24.xiii MBq | PR | Goserelin, Denosumab, Enzalutamide |
| seven yard/56 | Bone 3/3 | nd/−11.4 | 6 22.78 MBq | PR | Lupron, Denosumab, Abiraterone |
| 8 1000/58 | Bone 3/3 | nd/−43.0 | six 23.71MBq | SD/PD | Denosumab, Degarelix, Abiraterone |
| 9 m/sixty | Bone 2/3 | nd/−68.4 | half-dozen sixteen.45 MBq | SD | Buserelin, Denosumab, Enzalutamide |
| 10 m/83 | Bone 1/3 | +six.three/−63.5 | vi 24.06 MBq | PR | Denosumab, Degarelix, Enzalutamide |
Statistical analysis: The caused results were expressed as the mean ± SEM for each index. Comparing of data among various groups was performed with Student's unpaired t-test. A p < 0.05 was considered statistically significant. Correlation between groups was calculated using linear regression assay.
3. Results
The summary of imaging results is presented in Table two. All patients responded to their treatment according to the [18F]-NaF imaging data. Their PSA-curves are shown in Effigy 1. Fractional response, i.east., 25%–75% decrease in Southward-PSA values was seen 5 patients. One patient demonstrated consummate response, i.e., S-PSA values were = 0.0 or decreased >75%. Stable affliction <25% decrease or increase was seen in iii patients and i patient had progressive disease.
Figure one. PSA-curves before, during 6 cycles, and one month later on Ra-223 treatments in all patients; response, if >25% decrease in South-PSA values. SD = stable disease, PR = fractional response (>25% subtract), CR = complete response (>75% decrease), PD = progressive illness (>25% increase). The number refers to patient number (Tabular array ane and Table two), y-axis S-PSA concentration in μg/L, ten-axis fourth dimension in days, starting from the solar day of 1st Ra-223 treatment. Numbers one–x refer to patient number in Table 1, Table 2 .
Figure 1. PSA-curves earlier, during 6 cycles, and 1 month after Ra-223 treatments in all patients; response, if >25% decrease in S-PSA values. SD = stable disease, PR = partial response (>25% decrease), CR = complete response (>75% subtract), PD = progressive affliction (>25% increment). The number refers to patient number (Table 1 and Table 2), y-axis Southward-PSA concentration in μg/L, ten-centrality time in days, starting from the mean solar day of 1st Ra-223 handling. Numbers ane–10 refer to patient number in Table 1, Table 2 .

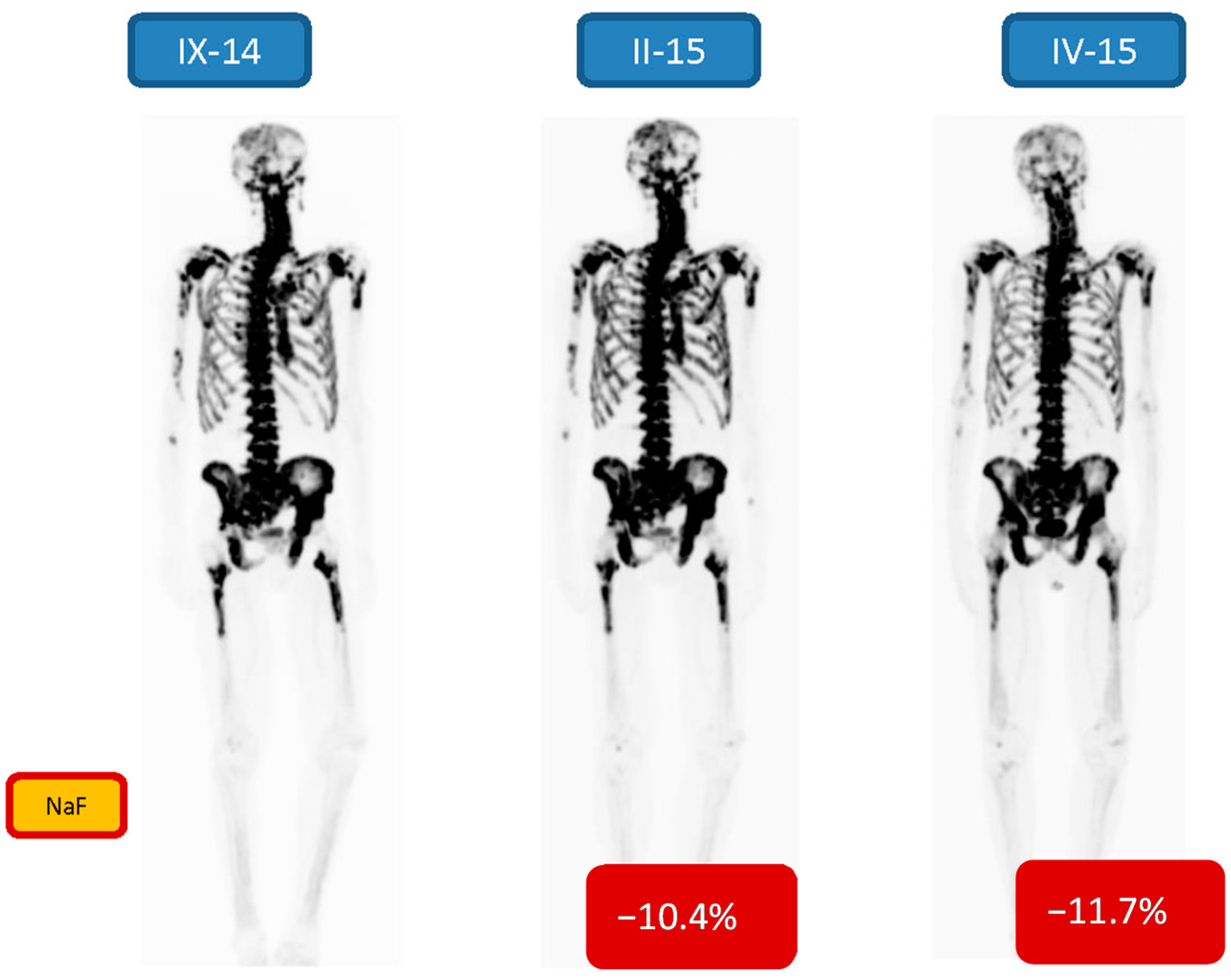
A few patient examples with imaging data are shown. Figure 2 demonstrates the [18F]-NaF imagings before, one interim assay (after 2 cycles) and one evaluation afterward the half dozen six cycles in a 69-year former male (patient no. 6). His Gleason score was viii (diagnosed in 2012), treated earlier with Casodex, Zoladex, PSA 671 μg/L at initial visit (in VII/14), and he was treated with 6 cycles of 223RaCl2. Three evaluations are shown: A [18F]-NaF-PET-CT before, one interim (after two cycles) and one afterward the six six cycles of Ra-223. There were quantitative changes (−10.4% and −11.7%) in both the interim and post therapy NaF-PET. The PSA-curve demonstrated fractional response (6, Figure i), only the ALP curve clearly demonstrated the delay in response due to 223RaCl2 production failure (interval between 1. and two. bike was x weeks).
Figure 2. 69-yr old male, Gleason score 8 (diagnosed in 2012), treated earlier with Casodex, Zoladex, Due south-PSA 671 ng/mL at initial visit in (Vii/14), was treated with half-dozen cycles of 223RaCl2. A NaF-PET-CT report before (9-14), one interim report (after two cycles, II-15) and one study (IV-15) after the 6 half dozen cycles are shown. There is a quantitative response (−10.iv% and −xi.7%) in both the interim and post therapy NaF-PET-maximum intensity projection images.
Figure 2. 69-twelvemonth old male person, Gleason score viii (diagnosed in 2012), treated earlier with Casodex, Zoladex, S-PSA 671 ng/mL at initial visit in (7/14), was treated with 6 cycles of 223RaCltwo. A NaF-PET-CT report before (Nine-xiv), i interim study (subsequently two cycles, II-15) and one report (Four-15) after the 6 six cycles are shown. In that location is a quantitative response (−x.iv% and −11.7%) in both the acting and post therapy NaF-PET-maximum intensity projection images.
Effigy three demonstrates some other patient (no. 2) with Gleason score seven treated with radical prostatectomy, (diagnosed in 2001), and with postoperative external beam radiation therapy, then with bicalutamide, docetaxel, abiraterone, enzalutamide, denosumab and goserelin. He demonstrated a biochemical relapse with a few findings on [18F]-FACBC-PET-CT [31], but with more than findings on [18F]-FCH-PET-CT iii months later. The response to the treatment was seen on [18F]-FCH-PET in 8 months, where the changes in the os marrow had dramatically disappeared. However, [18F]-NaF-PET on the following twenty-four hour period demonstrated a widespread active cortical os disease. This did not respond to the treatment after 1st wheel (+6.ii% change), but after 6 cycles a minor response was seen (−6.2% alter).
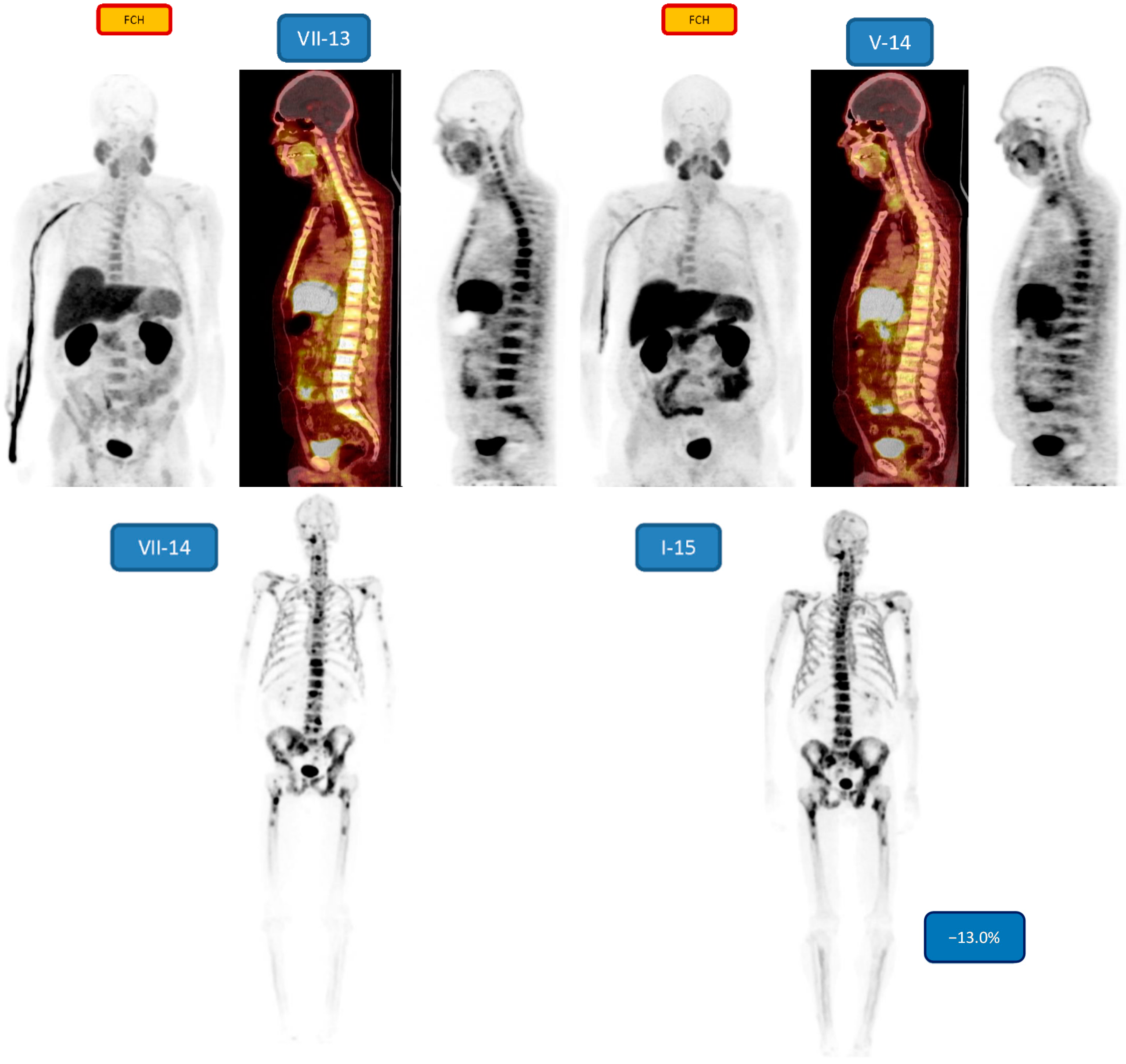
Figure 4 demonstrates a young patient (49 years, patient no. 4) with Gleason score nine, treated earlier with cyproterone, degarelix, bicalutamide, zoledronate, docetaxel, Sm-153-EDTMP, mitoxantrone, EBRT (P), docetaxel, abiraterone.
A response to the treatments is seen on [18F]-FCH–PET in 10 months, specially in the bone marrow disease.
[eighteenF]-NaF-PET half-dozen weeks later demonstrated a widespread active cortical bone disease. This responded to the Ra-223 treatment, considering afterward half-dozen cycles a response was seen (−thirteen.0% change).
Effigy 5 demonstrates an onetime patient (83 years) with Gleason score 6 (patient no. ten). The [18F]-NaF-PET before the Ra-223 treatment is shown, but no essential change in the full [eighteenF]-NaF-PET action due to the Ra-223 treatment was seen afterward 1st cycle (+half dozen.3% change), whereas after 6 cycles a dramatic response was seen (−63.5% change).
Figure 3. Demonstrates a patient with Gleason score 7 treated originally with radical prostatectomy in 2001, treated later with external beam radiations therapy, then with bicalutamide, docetaxel, abiraterone, enzalutamide, denosumab, doserelin. He demonstrated a biochemical relapse with a few findings on FACBC-PET (V-thirteen), just with more findings on FCH-PET three months later (VIII-13). The response to the handling was seen on FCH –PET in 8 months (IV-fourteen), where the changes in the bone marrow had dramatically disappeared. However, NaF-PET on the post-obit day (IV-xiv) demonstrated a widespread agile cortical bone disease. This did not respond to the treatment after 1st cycle (+6.2% modify, V-14), only after 6 cycles a response was seen (−6.2% change, IX-xiv).
Figure iii. Demonstrates a patient with Gleason score 7 treated originally with radical prostatectomy in 2001, treated after with external axle radiation therapy, then with bicalutamide, docetaxel, abiraterone, enzalutamide, denosumab, doserelin. He demonstrated a biochemical relapse with a few findings on FACBC-PET (V-13), but with more than findings on FCH-PET 3 months later on (Eight-13). The response to the handling was seen on FCH –PET in 8 months (IV-14), where the changes in the bone marrow had dramatically disappeared. Notwithstanding, NaF-PET on the following day (IV-14) demonstrated a widespread active cortical os disease. This did not respond to the treatment after 1st bike (+6.two% modify, V-14), but later on half-dozen cycles a response was seen (−vi.2% change, Ix-fourteen).
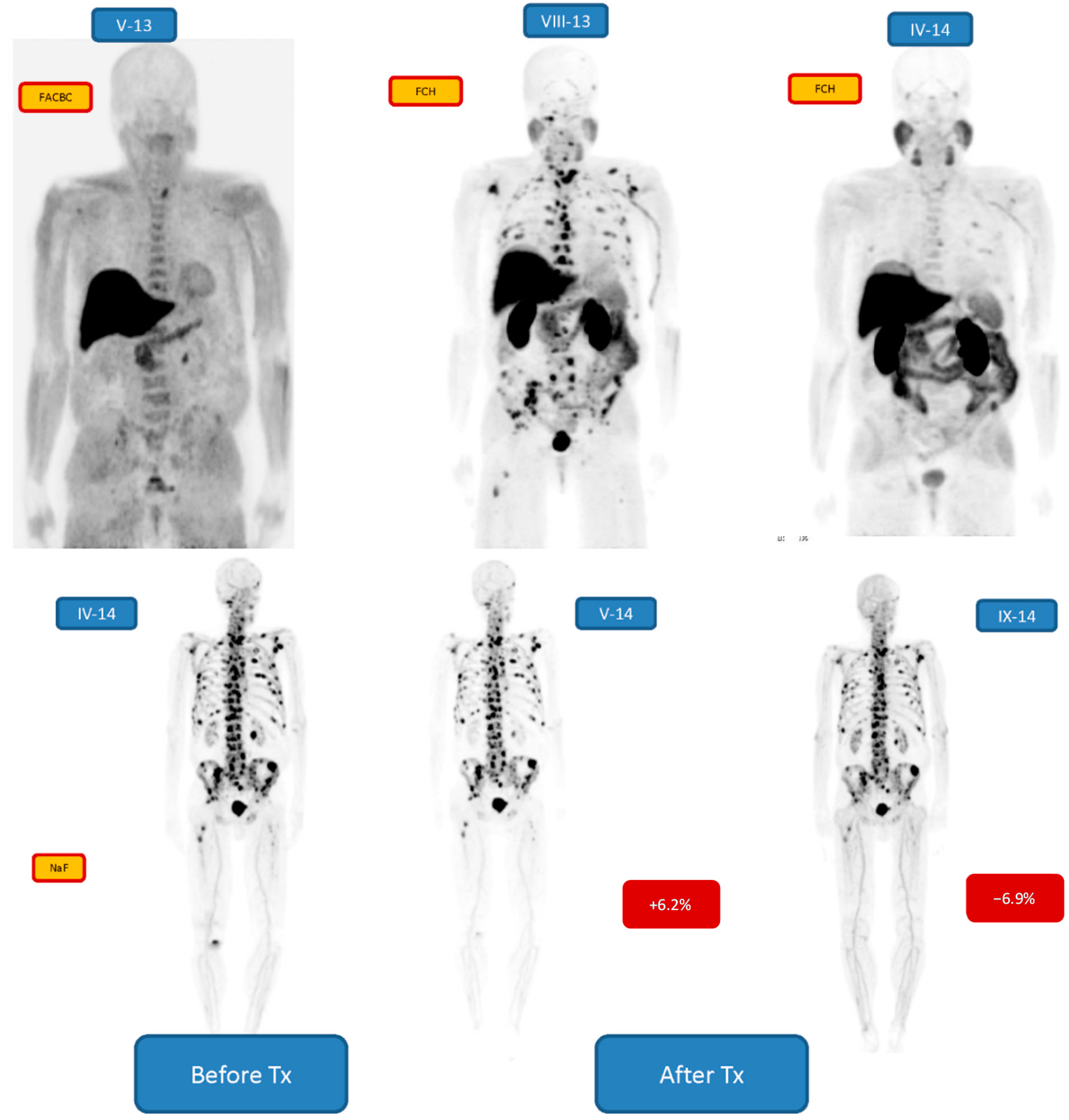
Figure 4. Demonstrates a young patient (49 years) with Gleason score nine, treated earlier with cyproterone, Degarelix, bicalutamide, zoledronate, docetaxel, Sm-153-EDTMP, mitoxantrone, EBRT (P), docetaxel, abiraterone. A response to the treatments is seen on FCH-PET in 10 months, especially in the bone marrow disease, MIP-image, sagittal fusion FCH-PET on CT and sagittal FCH-PET figures are shown in July 2013 (VII-13) and corresponding data in May 2014 (V-fourteen). NaF-PET 6 weeks (VII-14) later in July 2014 demonstrated a widespread active cortical os disease. This responded to the Ra-223 treatment, considering after 6 cycles a response was seen in January 2015 (−13.0% change, I-15).
Figure 4. Demonstrates a immature patient (49 years) with Gleason score 9, treated before with cyproterone, Degarelix, bicalutamide, zoledronate, docetaxel, Sm-153-EDTMP, mitoxantrone, EBRT (P), docetaxel, abiraterone. A response to the treatments is seen on FCH-PET in 10 months, especially in the os marrow affliction, MIP-epitome, sagittal fusion FCH-PET on CT and sagittal FCH-PET figures are shown in July 2013 (VII-xiii) and corresponding information in May 2014 (V-14). NaF-PET 6 weeks (Seven-fourteen) later in July 2014 demonstrated a widespread agile cortical bone disease. This responded to the Ra-223 treatment, because afterwards 6 cycles a response was seen in January 2015 (−13.0% alter, I-15).
Effigy 5. Demonstrates an onetime patient (83 years) with Gleason score 6. The NaF-PET before the Ra-223 handling half dozen cycles is shown (II-15), but no essential response to the treatment later 1st cycle (+vi.three% change, III-15) was seen, whereas after vi cycles a dramatic response was seen (−63.5% modify, VIII-xv). Many of the lesions have disappeared, especially in the thoracic girdle.
Figure 5. Demonstrates an old patient (83 years) with Gleason score 6. The NaF-PET before the Ra-223 treatment 6 cycles is shown (II-15), but no essential response to the treatment after 1st cycle (+6.3% change, III-15) was seen, whereas after half dozen cycles a dramatic response was seen (−63.5% modify, 8-15). Many of the lesions have disappeared, especially in the thoracic girdle.

iv. Discussion
Our patients with active mCRPC all responded to their treatments, considering quantitatively in that location was a pregnant decrease in [18F]-NaF-PET findings after 6 cycles of 223RaCl2 as compared to the baseline measurements. The quantitative decrement varied from −6.9% to −68.4%. The change tin can be visually seen in Effigy five (quantitatively −63.5%), but no then hands in Figure two, Figure 3 and Effigy four (quantitatively −6.9% to 13.0%). Therefore, quantification is important in widespread, i.e. more 10% of skeleton, metastatic bone disease. According to the S-PSA measurements all patients except one (patient no. i) showed some response in an active ambitious (T3-T4) widespread skeletal affliction.
There are at present multiple new therapies providing options for the treatment of patients having metastatic castration-resistant prostate cancer (mCRPC). For these patients, therapies were just very recently exceptionally ineffective and extremely limited to the utilize of docetaxel. These new therapies include androgen synthesis inhibitor (abiraterone) [32], androgen receptor signaling inhibitor (enzalutamide) [33], radionuclide therapy (radium-223) [34,35,36,37] and cabazitaxel chemotherapy [38].
Radium-223 was the first bone-seeking radionuclide that is reported to increase overall survival, and quality of life by providing pain relief in their late stage disease from symptomatic skeletal events (SSEs) such equally os pain, pathological fractures, or spinal cord compression seen in up to 90% of mCRPC patients. This new drug, radium-223-dichloride (223RaCl2) is a new bone-seeking calcium analogue alpha-emitter, offset of its kind in clinical use (Marketing authorization European union/i/xiii/873/001). It targets increased bone turnover adult by metastatic bone disease. In its double-blinded randomized registration trial, the 223RaCl2 receiving patients demonstrated median overall survival of xiv months, vs. 11.2 months for those on placebo [34,35].
Our patients with active mCRPC all responded to the given therapies. The common denominator was that they all received multimodality treatments with response. All of them received radium-223-dichloride (223RaCl2) plus denosumab, but otherwise this grouping of 10 patients was very heterogeneous. This is normally the instance in retrospective analysis describing the clinical do, when it comes to their demographics and previous and concomitant therapies (Tabular array 1 and Table two).
In add-on to the overall survival and QOL benefits, all the secondary endpoints were as well met in the Phase 3 ALSYMPCA (ALpharadin in SYMptomatic Prostate CAncer) registration trial [30,31,32,33], which included the delay in time to first skeletal-related events: first events occurred in 13.six months in the 223RaCl2 group every bit compared to 8.iv months in the placebo group—an improvement of 64%. 33% of 223RaCltwo patients had total alkaline phosphatase normalization equally compared to only one% in the placebo group. Additionally, the 223RaClii patients had an improvement of 49% in fourth dimension to PSA progression.
Very little, however, is known well-nigh the response evaluation of Ra-223 treatment. Although it has been shown that 223RaCl2 is a bone targeting treatment, its response using os targeting imaging has non yet been assessed in the literature. There is prove that NaF-PET can be used as a prognostic factor of the outcome of Ra-223 treatment [39]. They demonstrated that skeletal metastatic burden on baseline volition reflect on the outcome of 223RaClii treatment and advent of skeletal related events. In the PERCIST criteria [30] early minor response is recorded, if there is a 15% decrease in a total sum of v lesions; nevertheless, a maximum of two lesions can be taken from the same organ. In our modified criteria nosotros therefore selected just the ii lesions to stand for the whole skeleton. If we use the xxx% criteria for partial response as in PERCIST [30], the limit would be 12%, meaning that only five of our 10 patients demonstrated response. In the Medico Anderson Cancer Center written report [twoscore], also the total skeletal volume was analyzed based on SUV-threshold of 10. However, this cannot exist applied in the clinical routine, and information technology does not exclude heterogeneity in the metastatic distribution. Information technology is a challenge to place the best possible way of interpreting total malignant activity in the skeleton. In this work we wanted to present a elementary method for routinely analyzing the overall skeletal situation, and information technology seems, at least preliminarily, that we have been successful. The threshold has to be settled in a larger patient population.
All our patients were diagnosed with multiple PET tracers which all can be used for assessment of skeletal metastatic distribution. It is obvious, e.g., patient no. 2 who was scanned with [18F]-fluorocholine (FCH), [18F]-fluciclovine (FACBC) and [eighteenF]-NaF, that all these tracers requite different data most the in vivo biochemistry in prostate cancer. [18F]-FCH is an indicator for lipid metabolism and prison cell membrane synthesis in prostate cancer, [18F]-FACBC as an amino acid analog for protein synthesis and [18F]-NaF marking for an increased phosphate turnover in reactive osteoblastic cells. In our patients, os marrow uptake is typical to [18F]-FCH, cortical os uptake for NaF. The distribution for FCH and NaF can exist totally different, as shown in Figure three on studies on consecutive days.
Flare phenomenon might have been seen in 3 out of 6 patients (Figure 3 and Figure 5), which may indicate that the treatment response analysis after 1st cycle is not useful. The interim [xviiiF]-NaF uptake may besides be due to disease progression, but the S-PSA values practise non support this finding. At least, in this limited material at that place was no trend in the acting [eighteenF]-NaF values afterwards first or second wheel. The [eighteenF]-NaF uptake later on vi cycles, still, was significantly lower in all x patients equally compared to the baseline.
Therefore, we postulate that skeletal response to therapies can be assessed with [18F]-NaF. At that place was also a modify in PSA (Figure ane). Response to the therapies can be due to combined effect of abiraterone, enzalutamide or denosumab and Ra-223 alone. Ra-223 and denosumab were the only mutual regimens in all these therapies. On the other manus Ra-223 tin be combined with other therapies with success even though the mechanisms and combinations take to be studied carefully in order to sympathise the causality.
5. Determination
Our results suggest that eighteenF-Fluoride PET/CT is the nigh useful PET method in the follow-up of castration resistant prostate cancer with skeletal metastases. The presented quantification technique suits for response evaluation of 223RaCl2 in clinical routine.
Author Contributions
Both authors participated in all elements of the study (pattern, data collection, clinical evaluation etc.). Kalevi Kairemo was mainly responsible for imaging evaluations and quantification method development, whereas Timo Joensuu was mainly responsible for patient selection for Ra-223 and oncologic treatments. Both authors participated in manuscript writing.
Conflicts of Interest
Kalevi Kairemo has received consultancy fees from Bayer Healthcare AG (Wuppertal, Germany). Timo Joensuu is a shareholder of Docrates Cancer Center (Helsingfors, Finland).
References
- Wirth, M.; Horninger, West. How I treat metastatic prostate cancer. J. OncoPathol. 2014, 2, xiii–26. [Google Scholar] [CrossRef]
- Petrylak, D.P.; Tangen, C.Grand.; Hussain, Thousand.H.; Lara, P.N., Jr.; Jones, J.A.; Taplin, K.Eastward.; Burch, P.A.; Berry, D.; Moinpour, C.; Kohli, Yard.; et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N. Engl. J. Med. 2004, 351, 1513–1520. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Hotte, Due south.J. Guidelines for the management of castrate-resistant prostate cancer. Can. Urol. Assoc. J. 2010, 4, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Gleason, D.Chiliad.; Murray, R.; Tchekmedyian, Southward.; Venner, P.; Lacombe, L.; Chin, J.L.; Vinholes, J.J.; Goas, J.A.; Chen, B.; et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J. Natl. Cancer Inst. 2002, 94, 1458–1468. [Google Scholar] [CrossRef] [PubMed]
- Sathiakumar, N.; Delzell, E.; Morrisey, Yard.A.; Falkson, C.; Yong, One thousand.; Chia, V.; Blackburn, J.; Arora, T.; Kilgore, M.L. Mortality post-obit bone metastasis and skeletal-related events among men with prostate cancer: A population-based analysis of Us Medicare beneficiaries, 1999–2006. Prostate Cancer Prostatic Dis. 2011, 14, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Lange, P.H.; Vessella, R.L. Mechanisms, hypotheses and questions regarding prostate cancer micrometastases to bone. Cancer Metastasis Rev. 1998, 17, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Bodei, L.; Lam, M.; Chiesa, C.; Flux, M.; Brans, B.; Chiti, A.; Giammarile, F. European Association of Nuclear Medicine (EANM). EANM procedure guideline for treatment of refractory metastatic bone pain. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 1934–1940. [Google Scholar] [CrossRef] [PubMed]
- Pandit-Taskar, N.; Larson, S.Thou.; Carrasquillo, J.A. Bone-Seeking radiopharmaceuticals for treatment of osseous metastases, part 1: α Therapy with 223Ra-dichloride. J. Nucl. Med. 2014, 55, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, I. Strontium-89 for prostate cancer with bone metastases: The potential of cancer command and improvement of overall survival. Ann. Nucl. Med. 2014, 28, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Heron, D.Eastward.; Brufsky, A.; Beriwal, Due south.; Kurman, Thousand. Myelotoxicity of samarium Sm 153 lexidronam in patients receiving prior treatment with chemotherapy or radiotherapy. Ann. Oncol. 2008, xix, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Biersack, H.J.; Palmedo, H.; Andris, A.; Rogenhofer, S.; Knapp, F.F.; Guhlke, Due south.; Ezziddin, Due south.; Bucerius, J.; von Mallek, D. Palliation and survival after repeated 188Re-HEDP therapy of hormone-refractory os metastases of prostate cancer: A retrospective analysis. J. Nucl. Med. 2011, 52, 1721–1716. [Google Scholar] [CrossRef] [PubMed]
- Bilen, Thousand.A.; Johnson, Thousand.Thou.; Mathew, P.; Pagliaro, L.C.; Araujo, J.C.; Aparicio, A.; Corn, P.Yard.; Tannir, N.G.; Wong, F.C.; Fisch, M.J.; et al. Randomized stage 2 study of bone-targeted therapy containing strontium-89 in avant-garde castrate-sensitive prostate cancer. Cancer 2015, 121, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Harrison, M.R.; Wong, T.Z.; Armstrong, A.J.; George, D.J. Radium-223 chloride: A potential new treatment for castration-resistant prostate cancer patients with metastatic bone affliction. Cancer Manag. Res. 2013, 5, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sartor, O.; Coleman, R.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O'Sullivan, J.M.; Fosså, Southward.D.; Chodacki, A.; Wiechno, P.; Logue, J.; et al. Issue of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: Results from a phase 3, double-blind, randomised trial. Lancet Oncol. 2014, xv, 738–746. [Google Scholar] [CrossRef]
- Parker, C.; Nilsson, South.; Heinrich, D.; Helle, Due south.I.; O'Sullivan, J.M.; Fosså, Due south.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, G.; et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 2013, 369, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, M.; Fisher, D.R.; Roeske, J.C.; Bruland, O.Southward.; Larsen, R.H. Targeting of osseous sites with α-emitting 223Ra: Comparison with β-emitter 89Sr in mice. J. Nucl. Med. 2003, 42, 252–259. [Google Scholar]
- Nilsson, S.; Larsen, R.H.; Fosså, S.D.; Balteskard, L.; Borch, K.West.; Westlin, J.E.; Salberg, Chiliad.; Bruland, O.S. Showtime clinical feel with blastoff-emitting radium-223 in the handling of skeletal metastases. Clin. Cancer Res. 2005, xi, 4451–4459. [Google Scholar] [CrossRef] [PubMed]
- Segall, K.; Delbeke, D.; Stabin, Chiliad.One thousand.; Even-Sapir, Due east.; Fair, J.; Sajdak, R.; Smith, M.T. SNM practise guideline for sodium 18F-fluoride PET/CT bone scans. J. Nucl. Med. 2010, 51, 1813–1820. [Google Scholar] [CrossRef] [PubMed]
- Schirrmeister, H.; Guhlmann, A.; Elsner, Yard.; Kotzerke, J.; Glatting, G.; Rentschler, M.; Neumaier, B.; Trager, H.; Nussle, Thousand.; Reske, S.N. Sensitivity in detecting osseous lesions depends on anatomic localization: Planar bone scintigraphy versus 18F PET. J. Nucl. Med. 1999, 40, 1623–1629. [Google Scholar] [PubMed]
- Fifty-fifty-Sapir, E.; Metser, U.; Mishani, Eastward.; Lievshitz, Thou.; Lerman, H.; Leibovitch, I. The detection of os metastases in patients with high-risk prostate cancer: 99mTc-MDP Planar os scintigraphy, unmarried- and multi-field-of-view SPECT, 18F-fluoride PET, and 18F-fluoride PET/CT. J. Nucl. Med. 2006, 47, 287–297. [Google Scholar] [PubMed]
- Beheshti, M.; Vali, R.; Waldenberger, P.; Fitz, F.; Nader, Thousand.; Loidl, Due west.; Broinger, G.; Stoiber, F.; Foglman, I.; Langsteger, Westward. Detection of bone metastases in patients with prostate cancer by xviiiF fluorocholine and eighteenF fluoride PET-CT: A comparative study. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 1766–1774. [Google Scholar] [CrossRef] [PubMed]
- Withofs, N.; Grayet, B.; Tancredi, T.; Rorive, A.; Mella, C.; Giacomelli, F.; Mievis, F.; Aerts, J.; Waltregny, D.; Jerusalem, G.; Hustinx, R. 18F-fluoride PET/CT for assessing bone interest in prostate and breast cancers. Nucl. Med. Commun. 2011, 32, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Jadvar, H.; Desai, B.; Ji, 50.; Conti, P.Southward.; Dorff, T.B.; Groshen, Due south.Thousand.; Gross, M.E.; Pinski, J.K.; Quinn, D.I. Prospective evaluation of 18F-NaF and eighteenF-FDG PET/CT in detection of occult metastatic disease in biochemical recurrence of prostate cancer. Clin. Nucl. Med. 2012, 37, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Kjolhede, H.; Ahlgren, G.; Almquist, H.; Liedberg, F.; Lyttkens, K.; Ohlsson, T.; Bratt, O. Combined 18F-fluorocholine and 18F-fluoride positron emission tomography/computed tomography imaging for staging of high-chance prostate cancer. BJU Int. 2012, 110, 1501–1506. [Google Scholar] [CrossRef] [PubMed]
- Mosavi, F.; Johansson, S.; Sandberg, D.T.; Turesson, I.; Sorensen, J.; Ahlstrom, H. Whole-Body diffusion-weighted MRI compared with 18F-NaF PET/CT for detection of os metastases in patients with high-adventure prostate carcinoma. AJR Am. J. Roentgenol. 2012, 199, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
- Damle, North.A.; Bal, C.; Bandopadhyaya, G.P.; Kumar, L.; Kumar, P.; Malhotra, A.; Lata, S. The office of 18F-fluoride PET-CT in the detection of bone metastases in patients with chest, lung and prostate carcinoma: A comparison with FDG PET/CT and 99mTc-MDP os scan. Jpn. J. Radiol. 2013, 31, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Hillner, B.E.; Siegel, B.A.; Hanna, Fifty.; Duan, F.; Shields, A.F.; Coleman, R.E. Impact of 18F-fluoride PET in patients with known prostate cancer: Initial results from the National Oncologic PET Registry. J. Nucl. Med. 2014, 55, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Langsteger, Due west.; Balogova, S.; Huchet, V.; Beheshti, M.; Paycha, F.; Egrot, C.; Janetschek, K.; Loidl, W.; Nataf, V.; Kerrou, 1000.; et al. Fluorocholine (18F) and sodium fluoride (18F) PET/CT in the detection of prostate cancer: Prospective comparison of diagnostic performance determined by masked reading. Q. J. Nucl. Med. Mol. Imaging 2011, 55, 448–457. [Google Scholar] [PubMed]
- Von Eyben, F.E.; Kairemo, Thousand.; Kiljunen, T.; Joensuu, T. Planning of external beam radiotherapy guided by PET/CT. Curr. Radiopharm. 2015, 8, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Xofigo® g kBq/mL solution for injection. Summary of Product Characteristics. Bayer Pharma AG, Berlin, Germany. Revision dated November 2013. Available online: http://www.ema.europa.european union/docs/en_GB/document_library/EPAR_-_Product_Information/homo/002653/WC500156172.pdf (accessed on 6 Nov 2014).
- Wahl, R.50.; Jacene, H.; Kasamon, Y.; Society, Yard.A. From RECIST to PERCIST: Evolving considerations for PET response criteria in solid tumors. J. Nucl. Med. 2009, 50, 122S–150S. [Google Scholar] [CrossRef] [PubMed]
- Kairemo, K.; Rasulova, North.; Partanen, M.; Joensuu, T. Preliminary clinical experience of trans-ane-Amino-3-(eighteen)F-fluorocyclobutanecarboxylic acid (anti-(xviii)F-FACBC) PET/CT imaging in prostate cancer catients. Biomed. Res. Int. 2014, 2014, 305182. [Google Scholar] [CrossRef] [PubMed]
- Logothetis, C.J.; Basch, E.; Molina, A.; Fizazi, G.; North, S.A.; Chi, Chiliad.N.; Jones, R.J.; Goodman, O.B.; Mainwaring, P.N.; Sternberg, C.Due north.; et al. Effect of abiraterone acetate and prednisone compared with placebo and prednisone on hurting control and skeletal-related events in patients with metastatic castration resistant prostate cancer: Exploratory analysis of data from the COU-AA 301 randomised trial. Lancet Oncol. 2012, thirteen, 1210–1217. [Google Scholar] [PubMed]
- Scher, H.I.; Fizazi, Thou.; Saad, F.; Taplin, Grand.E.; Sternberg, C.N.; Miller, K.; de Wit, R.; Mulders, P.; Chi, Thousand.Due north.; Shore, N.D.; et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 2012, 367, 1187–1197. [Google Scholar] [PubMed]
- Nilsson, Southward.; Strang, P.; Aksnes, AK.; Franzèn, L.; Olivier, P.; Pecking, A.; Staffurth, J.; Vasanthan, Southward.; Andersson, C.; Bruland, Ø.S. A randomized, dose-response, multicenter phase II study of radium-223 chloride for the palliation of painful os metastases in patients with castration-resistant prostate cancer. Eur. J. Cancer 2012, 48, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, Southward.; Franzén, L.; Parker, C.; Tyrrell, C.; Blom, R.; Tennvall, J.; Lennernäs, B.; Petersson, U.; Johannessen, D.C.; Sokal, Grand.; et al. 2-Yr survival follow-up of the randomized, double-blind, placebo-controlled phase Ii study of radium-223 chloride in patients with castration-resistant prostate cancer and bone metastases. Clin. Genitourin. Cancer 2013, 11, twenty–26. [Google Scholar] [CrossRef] [PubMed]
- Humm, J.Fifty.; Sartor, O.; Parker, C.; Bruland, O.S.; Macklis, R. Radium-223 in the treatment of osteoblastic metastases: A critical clinical review. Int. J. Radiat. Oncol. Biol. Phys. 2015, 91, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Oyen, W.; Sundram, F.; Haug, A.R.; Kairemo, K.; Lewington, V.; Mäenpää, H.; Mortensen, J.; Mottaghy, F.; Virgolini, I.; O'Sullivan, J.Thou.; et al. Radium-223 dichloride (Ra-223) for the treatment of metastatic castration-resistant prostate cancer: Optimizing clinical practice in nuclear medicine center. J. OncoPathol. 2015, 3, 1–25. [Google Scholar] [CrossRef]
- Basch, Eastward.; Loblaw, D.A.; Oliver, T.K.; Carducci, M.; Chen, R.C.; Frame, J.Due north.; Garrels, K.; Hotte, S.; Kattan, Chiliad.Due west.; Raghavan, D.; et al. Systemic therapy in men with metastatic castration-resistant prostate cancer: American Order of Clinical Oncology and Cancer Care Ontario clinical practice guideline. J. Clin. Oncol. 2014, 32, 3436–3448. [Google Scholar] [CrossRef] [PubMed]
- Etchebehere, Eastward.C.; Araujo, J.C.; Fox, P.S.; Swanston, N.M.; Macapinlac, H.A.; Rohren, E.M. Prognostic factors in patients treated with Ra-223: The function of skeletal tumor burden on baseline 18F-fluoride-PET/CT in predicting overall survival. J. Nucl. Med. 2015, 56, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
© 2015 past the authors; licensee MDPI, Basel, Switzerland. This article is an open access commodity distributed nether the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Source: https://www.mdpi.com/2075-4418/5/4/413/htm
0 Response to "Radium-223 in the Treatment of Osteoblasticmetastases a Critical Clinical Review"
Post a Comment